In this short piece of article, we will see the key difference between isobars and isotopes in tabular form. So let’s get started…
What are isobars?
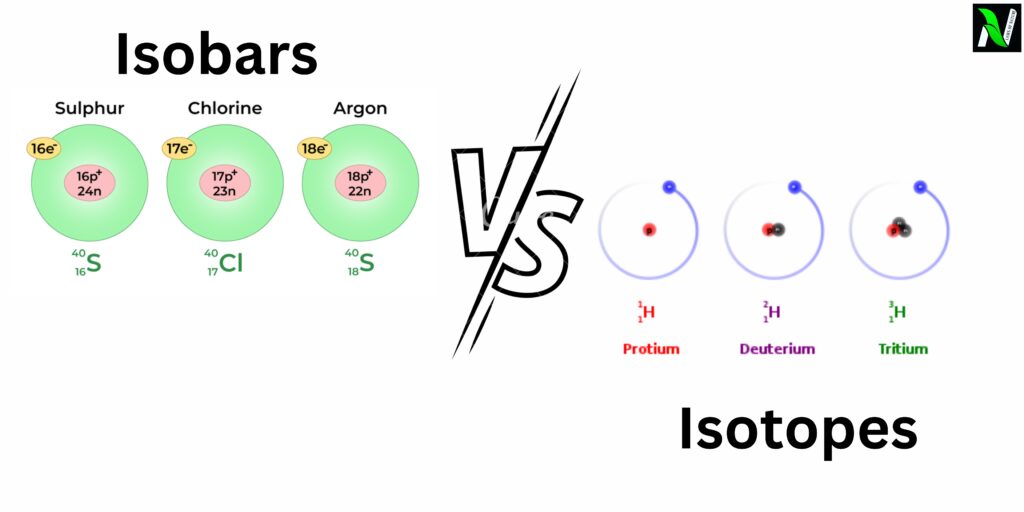
Isobars definition: Isobars are atoms of different elements that have the same mass number but different atomic numbers.
Example of isobar
Argon-40 (40Ar) and Calcium-40 (40Ca): Both have the same mass number of 40, but different atomic numbers. Argon-40 has 18 protons and 22 neutrons, while Calcium-40 has 20 protons and 20 neutrons.
What are isotopes?
Isotopes definition: Isotopes are atoms of the same element that have the same atomic number, but different mass numbers.
Example of isotope
Hydrogen Isotopes:
- Protium (1H): Contains a single proton and no neutrons.
- Deuterium (2H): Contains a single proton and one neutron.
- Tritium (3H): Contains a single proton and two neutrons.
Difference between isobars and isotopes in tabular form
Difference between isobars and isotopes in tabular form is given below:
| Criteria | Isotopes | Isobars |
|---|---|---|
| Definition | Atoms of the same element with the same atomic number (number of protons) but different mass number (number of protons + neutrons). | Different elements with the same mass number (total number of protons and neutrons) but different atomic numbers (number of protons). |
| Example | Carbon-12, Carbon-13, Carbon-14 | Carbon-14, Nitrogen-14 |
| Mass Number | May have slightly different physical properties due to different mass numbers. | Same among isobars. |
| Atomic Number | Same among isotopes of the same element. | Differs among isobars. |
| Chemical Properties | Similar chemical properties due to the same number of protons. | Different chemical properties due to different atomic numbers. |
| Position on the Periodic Table | Same element, so same position on the periodic table. | Different elements, so different positions on the periodic table. |
| Physical Properties | Dating artifacts, medical imaging, nuclear power, and understanding atomic reactions. | Not directly related to physical properties. |
| Applications | Dating artifacts, medical imaging, nuclear power, and understanding atomic reactions. | Understanding the diversity of elements, chemical reactions, and the periodic table. |
So, these are the difference between isobars and isotopes in tabular form, including their definitions, examples, mass numbers, atomic numbers, chemical properties, positions on the periodic table, physical properties, and applications.
Read Also
- Isobars and Isotopes
- Atomic number and atomic mass class 11
- Rutherford gold foil experiment class 11
- Rutherford atomic model: postulates, observations, and limitations, class 11
Stay tuned with Laws Of Nature for more useful and interesting content.